Optimization strategy of a new type of high-efficiency CO2 electroreduction composite catalyst
In recent years, with the proposal of the national "double carbon" development strategy, people pay more and more attention to the method of catalytic conversion of CO2 and synthesis of products with industrial value by using clean electric energy. Up to now, however, most of the research is still focused on the design and optimization of pure traditional solid catalysts or metal complex molecular catalysts, while there are few reports and studies on the composite catalytic system of solid polymer materials/metal complex molecules. Recently, researcher Liu Yingshuo (first author)/expert Wang Shuqing (correspondent author) from China Petrochemical Company and Professor Nie Weixuan (correspondent author) from the School of Engineering of West Lake University jointly wrote an article, focusing on the composite catalyst system with metal complex molecules embedded in controllable polymer materials, systematically summarizing and combing its design concept, catalytic mechanism of highly efficient catalytic CO2 reduction and the application and reports of this kind of catalytic materials in recent years, and putting forward some future applications and prospects of this new composite catalyst system.
This review firstly summarizes and compares the catalytic characteristics of traditional solid-state catalysts, molecular catalysts and polymer-catalyst composite systems synthesized by using molecular catalysts as building blocks in recent years. Although these kinds of catalyst materials have their own advantages in catalytic activity (solid catalyst), product selectivity (molecular catalyst) and structural novelty (molecular polymerization catalyst), they can't reproduce the catalytic active site structure of biological enzymes in vivo and their superior catalytic efficiency and activity.
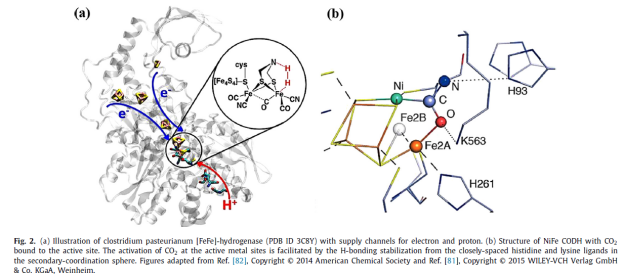
As shown in Figure 2, in biological enzymes, metal sites are used as catalytic substrates for binding/activating catalytic active sites (such as CO2 molecules), and the ligand structure connected with the metal center is used as the Primary Coordination Sphere, which can effectively regulate the catalytic activity of the active sites; In addition, the functional groups of protein structure around the metal sites, as the Secondary Coordination Sphere, can well stabilize the intermediates through the interaction with the active intermediates bound by the metal sites (for example, H bond interaction, electrostatic interaction, etc.), thus reducing the kinetic energy barrier of the catalytic process and improving the catalytic activity; The 3D protein structure can also provide a hydrophobic environment and construct an Outer Coordination Sphere through protein functional groups, thus reducing the occurrence of competitive reaction HER and improving the selectivity of CO2 reduction reaction.
Compared with the traditional solid-state catalysts, molecular catalysts and polymer-catalyst composite systems, Metal complex catalysts embedded in controllable polymer materials designed and developed by the author can well simulate the triple coordination environment of active sites in biological enzymes and realize efficient and selective catalytic reduction of CO2.
As shown in fig. 3, the molecular catalyst CoPc(cobalt phthalocyanine) is embedded in the polymer material P4VP(poly-4-vinylpyridine). In this structure, the Co center in CoPc is combined with Co2 molecules as a catalytic active site, and pyridine in P4VP can coordinate the CO center axially. As a Primary Coordination Sphere, the D-orbital energy level of the Co center is adjusted, the binding capacity of Co and CO2 is increased, and the catalytic activity is improved. At the same time, after the pyridine functional group suspended in P4VP is protonated, it can provide H bond interaction to stabilize the reduced CO2 active intermediate, thus serving as a Secondary Coordination Sphere to reduce the kinetic energy barrier of the catalytic process.
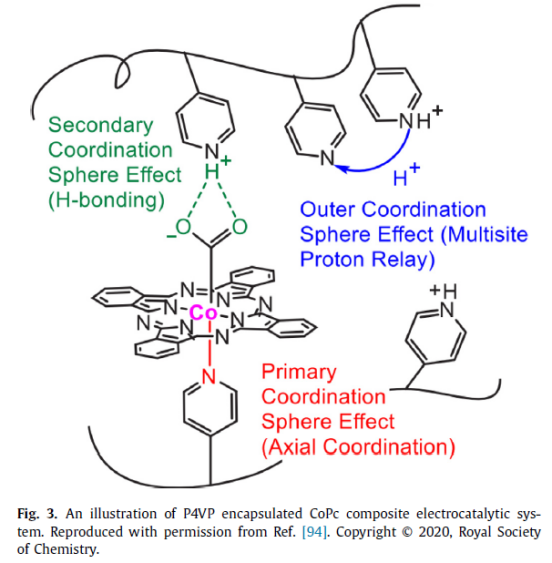
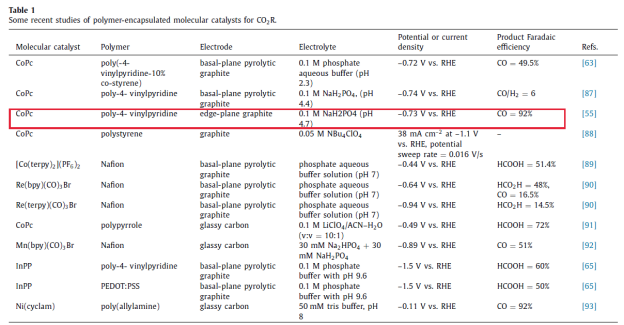
In addition, the hydrophobic structure of P4VP can effectively reduce the combination of H2O molecules and Co centers and inhibit the competitive reaction of HER. In neutral buffer solution, the pyridine functional group in P4VP will be partially protonated according to the pH environment of the solution, and such a special structure (Outer Coordination Sphere) can well transfer the necessary proton source for the CO2 reduction process, promote the CO2 reduction while ensuring the high selectivity of CO2 reduction, which perfectly reproduces the catalytic characteristics of bioactive enzymes in Figure 2. As can be seen from Table 1, compared with other catalytic systems, the CoPc/P4VP composite catalytic system shows higher catalytic activity and selectivity.
Polymer-encapsulated metal complex catalysts with metal complex molecules embedded in polymer materials are composed of metal complex molecules and polymer materials respectively, which also provides more ways and angles for regulating and optimizing their catalytic properties:
(1) Optimizing the selection of appropriate central metals: Through the reasonable selection of central metal ions, the activity of molecular catalysts for CO2 binding can be further optimized, and at the same time, the binding capacity for H+ can be reduced, and the selectivity of the catalytic system for CO2 reduction can be further optimized.
(2) Optimizing the modification of the structure of macrocyclic ligand: for example, adjusting the electrochemical properties of the ligand by introducing electron-withdrawing groups; Introducing charged groups to increase intramolecular electrostatic field effect; And increase the conjugated structure of macrocyclic ligands in order to regulate the activity, catalytic potential, structural stability and selectivity to specific products of molecular catalysts.
(3) Optimize and adjust the structure and characteristics of polymer: Because the suspended functional groups in the polymer structure can coordinate with the metal center in the molecular complex, the electron density and catalytic activity of the metal center can be regulated), the catalytic activity of the catalytic system can be regulated by adjusting the different electron donor capabilities of the suspended functional groups.
In addition, adjusting the conductivity of polymer/mixed materials, such as adding graphene carbon materials with good conductivity to improve the conductivity of non-conductive P4VP, can further enhance the catalytic activity of the catalytic system. At the same time, the regulation of the proportion of these solid mixtures provides another dimension of optimization strategy.
Source:https://mp.weixin.qq.com/s/K4fCbF21CohurCgn_fy2oQ
