Dual-Atom Relay Catalysis Enables Propane Combustion Below 200 °C: A Breakthrough for Efficient VOC Abatement
A breakthrough study recently published online in Nature Chemistry reports a highly efficient strategy for low-temperature catalytic combustion of propane using a current-assisted dual-atom catalyst. The research introduces a Pt–Nb dual-atom catalytic system supported on antimony-doped tin oxide (ATO), achieving complete propane conversion at temperatures below 200 °C (T₉₀ < 200 °C). At 220 °C, the catalyst reaches a turnover frequency as high as 27.67 × 10⁻³ s⁻¹ while reducing platinum usage by more than 80%.
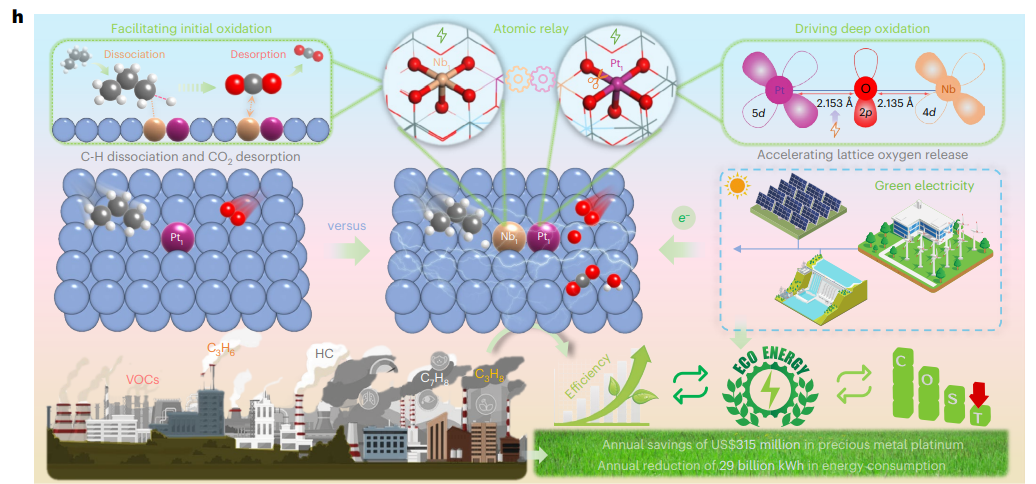
Through comprehensive in-situ characterization and theoretical simulations, the research team uncovered a current-assisted “atomic relay” mechanism underlying the catalyst’s exceptional activity. In this mechanism, Nb sites facilitate C–H bond activation in propane and promote CO₂ desorption, while the applied current weakens Pt–O bonding, accelerating the activation and release of lattice oxygen. This synergistic design provides a new paradigm for the low-temperature elimination of volatile organic compounds (VOCs).
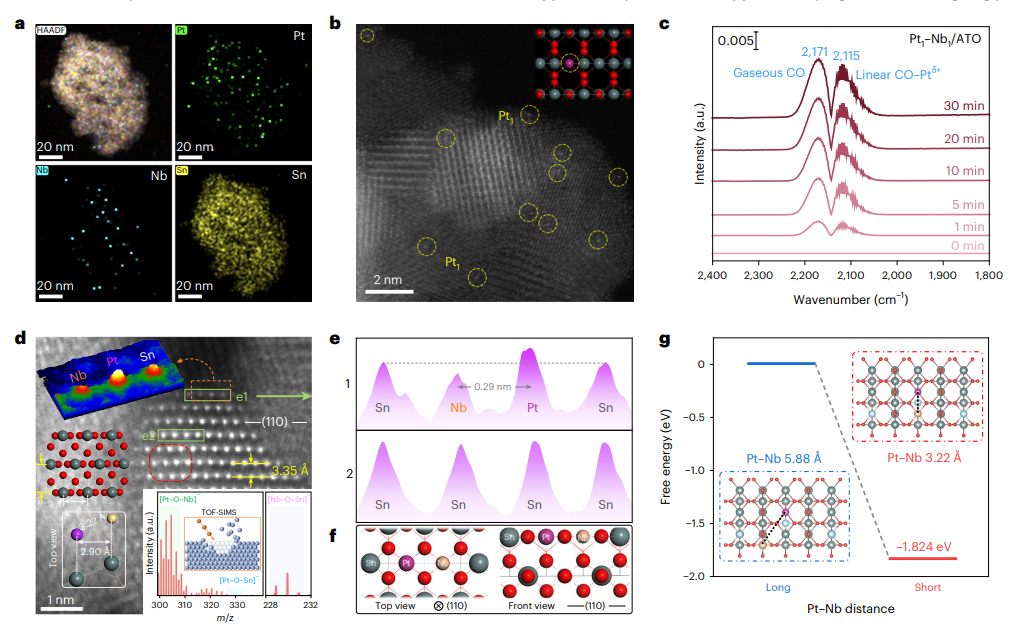
Extensive structural characterization confirmed the successful construction of the catalyst. X-ray diffraction and nitrogen adsorption–desorption measurements show that the catalyst retains the tetragonal SnO₂ structure with mesoporous characteristics. Surface analysis reveals that platinum primarily exists as oxidized Pt²⁺ species. Advanced electron microscopy demonstrates that Pt and Nb atoms are uniformly dispersed across the ATO support, with atomically dispersed Pt sites clearly visible at the atomic scale. Spectroscopic analysis further confirms that Pt is present predominantly in isolated atomic form rather than in clusters or nanoparticles.
More importantly, multiple lines of evidence reveal the formation of adjacent Pt–O–Nb dual-atom structures on the catalyst surface. Atomic-resolution imaging indicates that some surface Sn atoms in the SnO₂ lattice are replaced by Pt and Nb atoms, creating local atomic configurations that bring the two metals into close proximity. X-ray absorption spectroscopy provides deeper insight into the coordination environment, confirming Pt–O coordination without detectable Pt–Pt interactions, consistent with single-atom dispersion. Quantitative analysis shows that most Pt atoms are paired with neighboring Nb atoms, forming Pt₁–Nb₁ atomic pairs. Density functional theory calculations further demonstrate that forming these paired sites is thermodynamically more favorable than maintaining isolated Pt or Nb atoms.
To evaluate catalytic performance, the researchers developed a custom current-assisted catalytic reaction system in which the catalyst is positioned between conductive filters and connected to a controllable DC power supply. The applied current dramatically enhances catalytic performance. Under current assistance, the Pt₁–Nb₁/ATO catalyst achieves a T₅₀ of 140 °C and a T₉₀ of 192 °C, far outperforming conventional thermocatalytic conditions as well as catalysts containing only isolated Pt atoms. Decoupling analysis indicates that non-thermal effects induced by the electric current account for approximately 74% of the observed activity enhancement, confirming the dominant role of current-driven electronic effects rather than simple Joule heating.
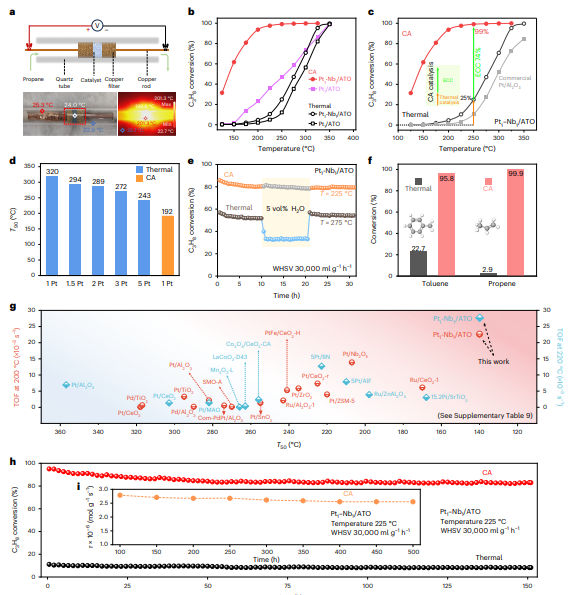
The catalyst also demonstrates remarkable efficiency in terms of precious-metal utilization. With only 1 wt% Pt loading under current-assisted conditions, the catalyst outperforms traditional thermocatalysts containing 5 wt% Pt, highlighting the potential to reduce noble-metal consumption by over 80%. Structural characterization after reaction confirms that the atomically dispersed Pt sites remain stable without aggregation, indicating excellent durability.
The system also exhibits outstanding hydrothermal stability. Even in the presence of 5 vol% water vapor, propane conversion remains unchanged under current-assisted conditions, whereas conventional thermocatalysis shows significant activity loss. Isotopic labeling experiments reveal that water molecules readily dissociate on the catalyst surface under current assistance and actively participate in propane oxidation rather than blocking active sites, thereby enhancing catalytic robustness.
Comparisons with previously reported catalysts highlight the outstanding performance of the system. The Pt₁–Nb₁/ATO catalyst achieves a T₅₀ as low as 140 °C and extremely high turnover frequencies at 200 °C and 220 °C, positioning it among the most active catalysts reported for propane combustion. Long-term testing further demonstrates stable activity over 500 hours without noticeable deactivation.
To understand the structural requirements behind this performance, the research team systematically synthesized catalysts with different active-site configurations. Catalysts containing randomly distributed Pt and Nb atoms, aggregated Pt clusters, excessive Nb loading, or large Pt–Nb separations all showed inferior activity. These results confirm that closely neighboring Pt–Nb dual-atom sites are essential for optimal catalytic performance. Increasing the average distance between Pt and Nb sites leads to a gradual decline in activity, highlighting the critical role of atomic-scale proximity in enabling cooperative catalysis.
Theoretical calculations reveal how the applied electric current modifies the catalyst’s electronic structure. Under current-induced charge conditions, the Pt–O bond length increases significantly, making it easier for lattice oxygen to be released. Electronic-structure analysis shows that the d-band centers of both Pt and Nb shift closer to the Fermi level, promoting oxygen activation and C–H bond cleavage. The coupling between oxygen 2p orbitals and Pt 5d orbitals becomes weaker, enhancing the electrophilicity of oxygen species and facilitating oxidation reactions.
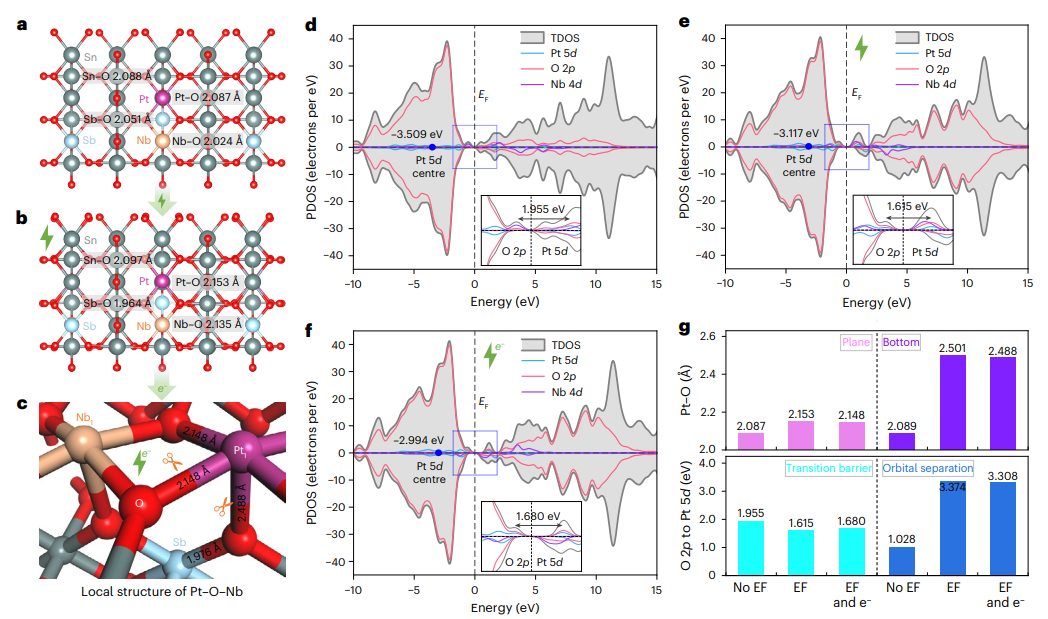
Based on these findings, the researchers propose a current-assisted atomic relay mechanism. In this process, Nb sites function as Lewis acid centers that promote C–H bond cleavage in propane and facilitate CO₂ desorption through σ-donation and d-back-donation interactions. Meanwhile, electron-enriched Pt sites activated by the electric current enable low-temperature activation of lattice oxygen and drive deep oxidation of reaction intermediates. The applied current thus acts as a targeted external stimulus, orchestrating a stepwise catalytic relay that dramatically enhances propane combustion efficiency at low temperatures.
The optimized Pt₁–Nb₁/ATO catalyst demonstrates exceptional catalytic performance, including a T₉₀ of only 192 °C, a turnover frequency of 27.67 × 10⁻³ s⁻¹ at 220 °C, and a low apparent activation energy of 41.2 kJ mol⁻¹. In addition to excellent hydrothermal stability and long-term durability, the catalyst also shows strong activity for the catalytic combustion of various light alkanes and aromatic VOCs such as toluene, indicating broad applicability.
The economic and environmental implications of this technology are substantial. By dramatically reducing noble-metal requirements and enabling efficient low-temperature operation, the strategy could save an estimated $315.54 million annually in precious-metal costs or reduce energy consumption by approximately 29.83 billion kilowatt-hours.
More broadly, the study demonstrates the transformative potential of current-assisted dual-atom catalysis for low-temperature oxidation reactions. By integrating external electric fields with atomically engineered active sites, this approach opens new avenues for electrically driven, intelligent catalytic systems. Beyond providing an efficient solution for VOC abatement, the concept of externally controlled atomic relay catalysis may inspire future catalyst designs capable of overcoming thermodynamic limitations and enabling highly efficient chemical transformations under mild conditions.
