Fluorine-Assisted Flash Joule Heating Enables Morphology-Controlled Silicon Carbide Nanowires with 71% Improved Wear Resistance
On March 10, 2026, researchers from Rice University, including Yi Cheng, Yufeng Zhao, and James M. Tour, reported a new strategy for synthesizing morphology-controlled carbide nanomaterials in a study published in Matter. The work introduces a fluorine-assisted flash Joule heating (FAF) approach capable of rapidly converting silica-rich waste—such as discarded glass—into silicon carbide (SiC) nanowires within seconds.
By introducing trace amounts of fluorine (~1 wt%), the researchers significantly enhanced the catalytic activity of intrinsic iron oxides present in waste glass. This activation enables selective nanowire growth with a nanowire selectivity of 78% and a silicon yield of 96%. Beyond SiC, the strategy can also be extended to other ultrahigh-temperature ceramic nanowires, including B₄C, TiC, and NbC, providing a sustainable pathway for transforming waste resources into high-value advanced materials.
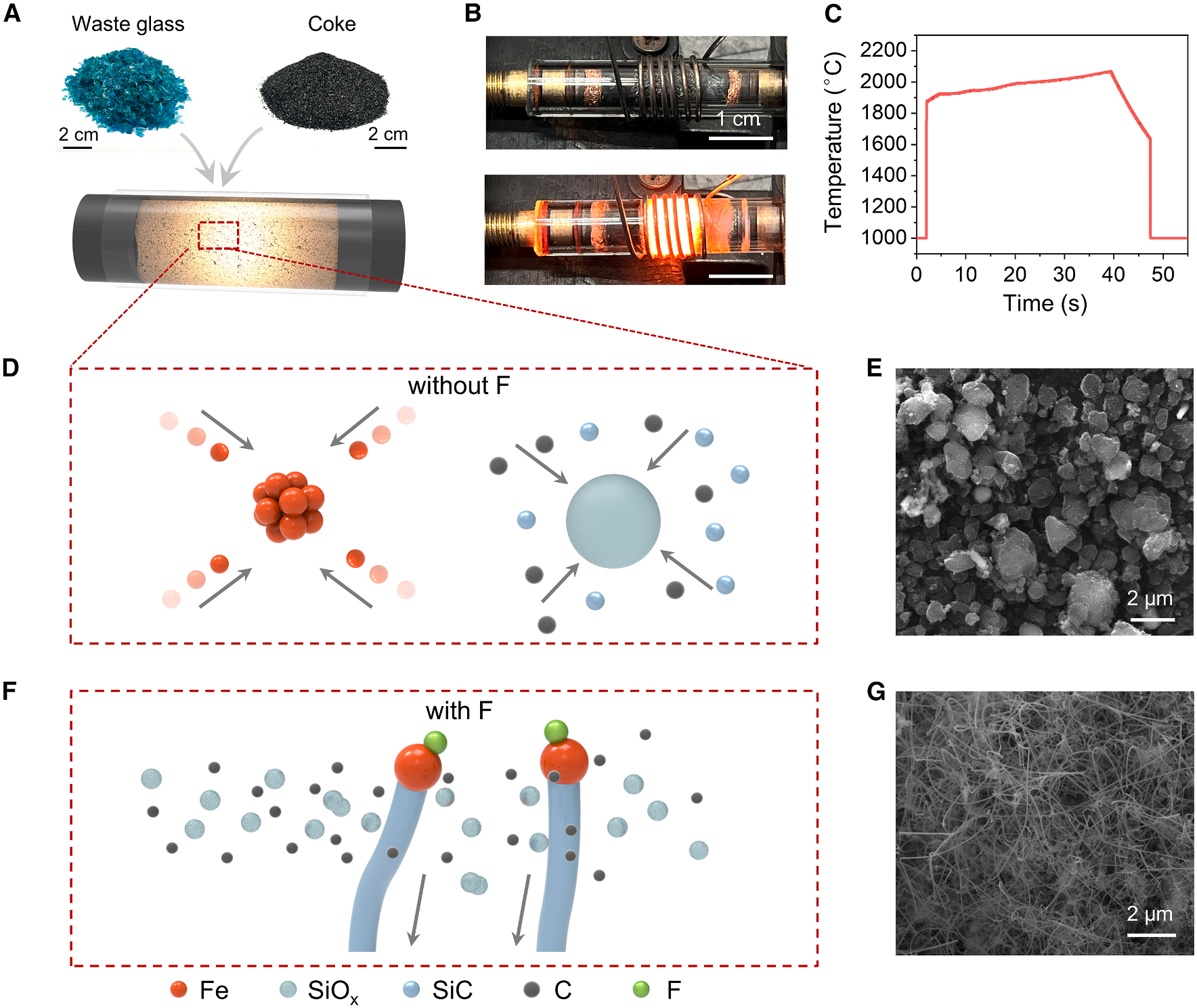
The synthesis relies on a rapid flash Joule heating process. Waste glass powder is mixed with carbon black and sodium fluoride (NaF) and loaded into a quartz reaction tube. Using a programmable power supply, an electrical current of 40 A is applied, rapidly heating the system to approximately 2000 °C within seconds. The reaction proceeds under extremely fast thermal cycles, with heating rates exceeding 10³ °C s⁻¹ and cooling rates greater than 10² °C s⁻¹, completing the transformation in just 30 seconds.
By tuning both heating duration and fluorine concentration, the researchers achieved precise control over nanowire morphology, including their diameter and length. Comprehensive characterization using X-ray diffraction, Raman spectroscopy, electron microscopy, and surface analysis confirmed the crystalline structure, morphology, and chemical composition of the resulting SiC nanowires. Under optimized conditions—30 seconds of heating and ~1 wt% fluorine—the nanowires exhibit diameters of about 70 nm and lengths approaching 20 μm.
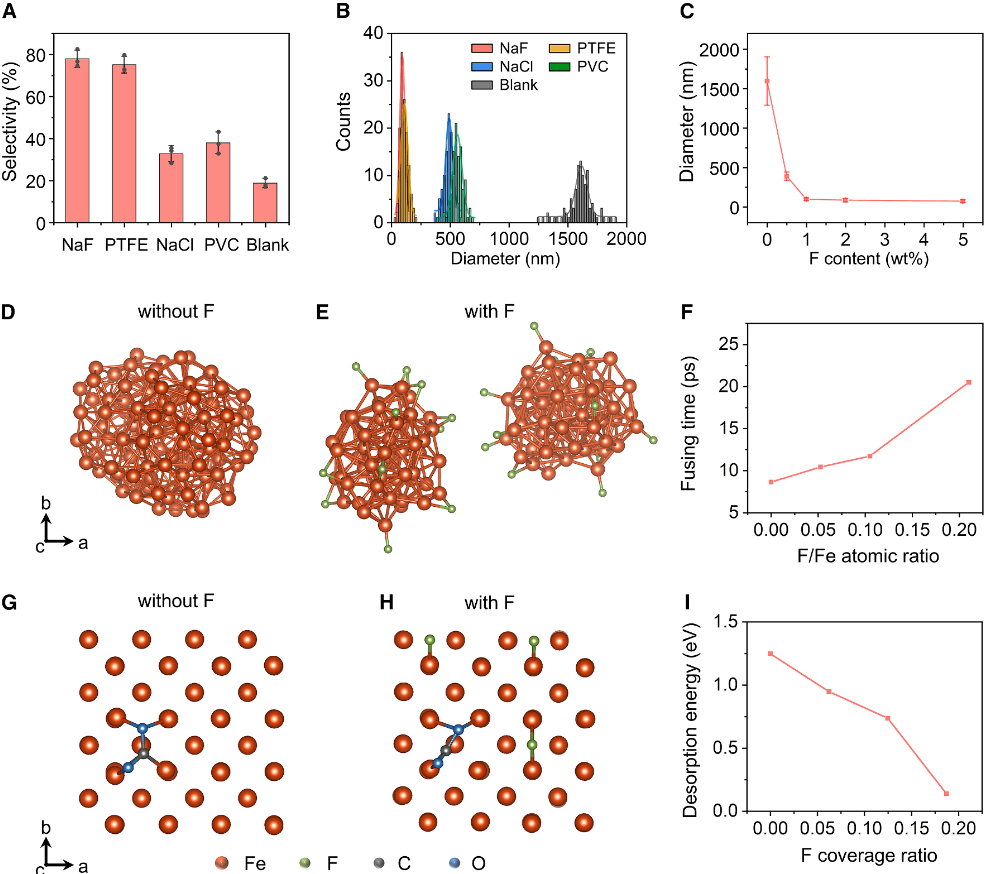
The study reveals that fluorine plays a decisive role in directing nanowire growth. In the absence of fluorine, the reaction primarily produces SiC particles. However, when NaF is introduced, the material undergoes oriented growth, forming one-dimensional nanowires. Molecular dynamics simulations and density functional theory calculations suggest that fluorine atoms adsorbed on the surface of iron catalysts suppress particle coalescence at high temperatures, extending the catalyst lifetime. At the same time, fluorine dramatically lowers the energy barrier for CO₂ desorption—from 1.25 eV to 0.14 eV—facilitating rapid regeneration of active sites and promoting continuous nanowire formation.
The resulting SiC nanowires demonstrate remarkable reinforcement effects when incorporated into polymer composites. When only 1 wt% of SiC nanowires is added to a vinyl ester resin matrix, the thermal conductivity increases from 0.17 to 0.31 W·m⁻¹·K⁻¹. Mechanical performance improves substantially as well, with Young’s modulus increasing by 41%, hardness by 62%, and wear rate reduced by 71%. In contrast, adding the same amount of SiC particles produces minimal improvement in thermal conductivity and reduces wear by only 53%. The superior performance of the nanowire-reinforced composites is attributed to the nanowires’ high aspect ratio, large surface area, and enhanced load-bearing capability.
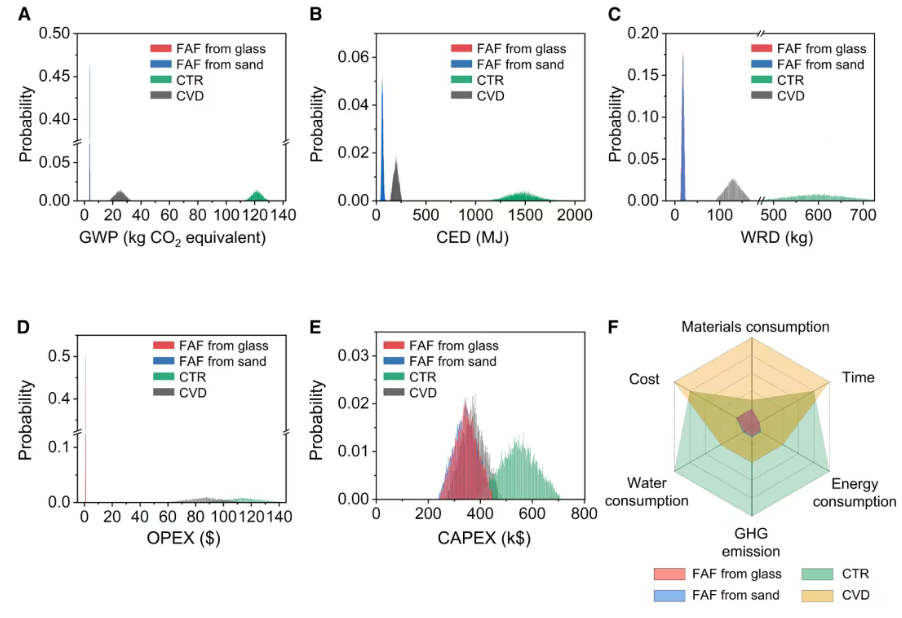
In addition to its materials performance, the FAF synthesis route also demonstrates strong environmental and economic advantages. Life-cycle assessment and techno-economic analysis show that the process reduces global warming potential to approximately 3.9 kg CO₂-equivalent per kilogram of SiC nanowires—an 85–96% reduction compared with conventional methods such as chemical vapor deposition or carbothermal reduction. The cumulative energy demand is reduced by 68–96%, while water consumption decreases by up to 97%. Operational costs drop dramatically to about $0.9 per kilogram of product, representing a 99% reduction compared with traditional processes. Capital expenditures are also reduced by 6–37% at a production capacity of 1 kg per hour.
The fluorine-assisted flash Joule heating strategy also demonstrates strong versatility. The research team successfully extended the approach to the synthesis of several other ultrahigh-temperature ceramic nanowires, including boron carbide, titanium carbide, and niobium carbide. This versatility suggests that the method could serve as a general platform for producing a wide range of one-dimensional ceramic nanomaterials.
Looking ahead, the researchers envision expanding the fluorine-assisted strategy to additional carbide and nitride systems while deepening the mechanistic understanding of fluorine’s catalytic role through in-situ characterization and multiscale modeling. Early scale-up experiments indicate that increasing the reaction scale can reduce unit energy consumption by nearly an order of magnitude. Combined with prior successes in ton-scale production of graphene using flash Joule heating technology, the FAF approach holds strong promise for industrial-scale manufacturing of SiC nanowires.
By transforming waste materials into high-performance nanomaterials through an ultrafast, energy-efficient process, this work highlights a new pathway toward sustainable materials manufacturing. The ability to simultaneously control nanostructure morphology, reduce environmental impact, and enhance composite performance positions fluorine-assisted flash Joule heating as a promising technology for future applications in aerospace, electronics, and advanced composite materials.
