Acid humidified carbon dioxide gas input improves CO2RR stability
Using renewable energy to electrochemically convert CO2 into valuable products is a promising technology for carbon utilization. Zero-gap membrane electrode assembly (MEA) electrolyzer stands out because of its high efficiency of CO2 reduction reaction. It integrates gas diffusion electrode (GDE) and anion exchange membrane (AEM), which can achieve high Faraday efficiency and is a strong candidate for practical application.
However, the commercialization of CO2RR MEA electrolyzer mainly has the following problems:
1. The long-term stable operation of CO2RR MEA electrolyzer is limited by the salt precipitation in cathode chamber.
The formation of salt in cathode chamber is the key problem of CO2RR MEA electrolyzer. Bicarbonate or carbonate crystals precipitate in GDE and gas flow channel, which hinders CO2 gas flow and causes overflow, reduces CO₂RR selectivity, and finally leads to electrolytic cell failure.
2. Further innovation is needed to promote the practical application of CO2RR MEA electrolyzer.
Advanced characterization techniques (such as X-ray diffraction, absorption, optical coherence tomography and neutron imaging) reveal the mechanism of salt formation, and stimulate many methods to reduce salt formation (such as adhesive engineering, pulse electrolysis, electrolyte selection, GDE reprocessing and membrane modification). However, these methods still face stability problems or unstable operation, and further innovation is needed to promote the practical application of CO2RR MEA electrolyzer.
In view of this, Professor Wang Haotian of Rice University in the United States and others have proved that the accumulation of salt can be prevented by introducing CO2 gas into the acid bubbler, which brings a small amount of acid vapor into the gas diffusion electrode of silver-catalyzed CO2RR to carbon monoxide (CO). In a 100-square-centimeter single serpentine channel CO2RR membrane electrode electrolyzer, the acid humidification method achieved a stable operation for 4500 hours at a current density of 100 mA cm-2 without affecting the Faraday efficiency of CO, while the conventional water humidification CO2 feed only operated stably for about 80 hours. The method of humidifying CO2 with acid can be extended to bismuth, copper and zinc catalysts.
Technical scheme
1. The degree of salt formation of CO2 humidified by H2O and HCl was observed and compared.
The traditional water humidified CO2RR electrolyzer has poor stability due to salt accumulation. Using volatile acid (such as HCl) to humidify CO2 can significantly reduce salt precipitation and prolong the stability of electrolytic cell to more than 1000 hours.
2. The mechanism that acid humidification significantly prolonged the electrolytic stability of CO2RR was explored.
It is found that acid humidification significantly reduces the formation and accumulation of salt in CO2RR electrolyzer and prolongs the stability to 2000 hours. Acid steam can increase the solubility of salt, reduce salt blockage and improve the performance of electrolyzer.
3. Various acids were used to screen the stability of CO2RR humidification.
The research shows that acid humidification significantly reduces the salt accumulation in CO2RR electrolyzer and prolongs the stability to more than 2000 hours. Low concentration acid can effectively remove salt without affecting catalytic performance. This method is suitable for various catalysts and AEM, and has wide applicability.
4. The stability of acid humidification method in 100 cm² CO2RRMEA electrolyzer was evaluated.
The author confirmed that in a 100 cm² MEA electrolyzer, acid humidification significantly prolonged the CO2RR stability to 2000-4500 hours, far exceeding the 80 hours of water humidification, and kept high CO Faraday efficiency.
Technical advantages
1. Innovative use of volatile acid solution to humidify CO2 gas flow.
In this paper, volatile acid solutions (such as HCl, HCOOH and CH3COOOH) are innovatively used to humidify the CO2 gas flow, which significantly reduces the formation of salt crystals in the cathode chamber, and the effect is outstanding compared with that of standard H2O humidification, and the optimized acid concentration has no negative impact on the CO Faraday efficiency and cell pressure of CO₂RR.
2. CO2RR with high stability and expansibility is realized by humidifying acid solution.
The CO2 gas humidified by HCl was electrolyzed continuously for more than 2000 hours at a current density of 100 mA cm-2, and the CO FE content was kept above 90%, which was successfully applied to a 100 cm² MEA electrolyzer. In addition, this method also significantly improves the stability of other catalysts, and there is no serious overflow risk, showing a wide range of applicability.
Technical details
1. Observation and comparison of CO2 humidified with H2O and HCl to form salt
The stability of CO₂RR was tested by continuous electrolytic operation. It was found that the MEA electrolyzer was significantly degraded within about 200 to 500 hours when CO₂ was humidified by traditional water. The main reason was that a large amount of potassium bicarbonate accumulated in the gas flow channel and the back of GDE, which hindered the CO₂ gas flow. In order to solve this problem, the author puts forward a method of humidifying CO₂ with volatile acid (such as HCl). The experiment shows that the CO₂ humidified by 0.1 M HCl not only maintains the catalytic activity similar to that of water humidification, but also significantly prolongs the stability of the electrolyzer to more than 1000 hours, and the Faraday efficiency of CO remains at a high level. Through the observation of a customized transparent gas flow plate electrolyzer, it is found that HCl humidification can effectively prevent the salt from accumulating in the gas flow channel, while water humidification can lead to the rapid accumulation of salt crystals. These results show that the acid wet method is an effective strategy, which can significantly reduce salt precipitation and improve the long-term operation stability of CO₂RR electrolyzer.
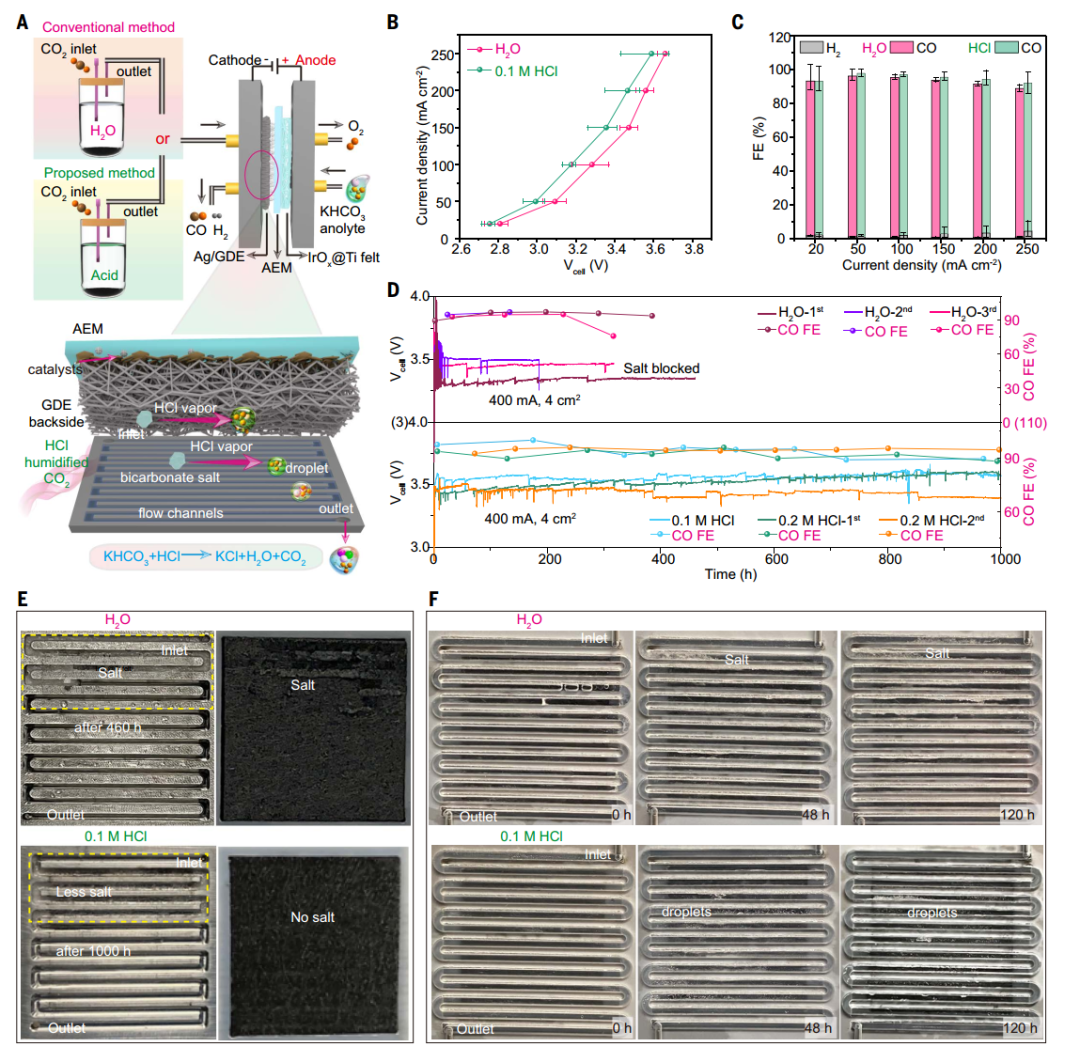
Fig. 1 Comparison of salt formation in MEA electrolyzers using H2O and acid humidified CO₂
2. Mechanism understanding
The author explored the reason why acid humidification significantly prolonged the electrolytic stability of CO₂RR through 1 cm² MEA electrolyzer. It was observed by optical microscope that salt crystals quickly accumulated on the back of GDE and gas flow channel when water was humidified, while salt formation and accumulation were significantly reduced when acid was humidified. SEM and EDS analysis showed that the number and size of salt crystals decreased under the condition of acid humidification. The experiment shows that the stability of electrolyzer is extended to 2000 hours under the condition of acid humidification, while the water humidification is only about 960 hours. X-ray diffraction analysis shows that KHCO3 crystal is not found in the flow channel during acid humidification, but KHCO3 is the main salt component in the liquid at the outlet of cathode chamber, which shows that acid vapor can effectively reduce salt blockage and significantly improve the stability of electrolytic cell by increasing salt solubility and promoting salt discharge.
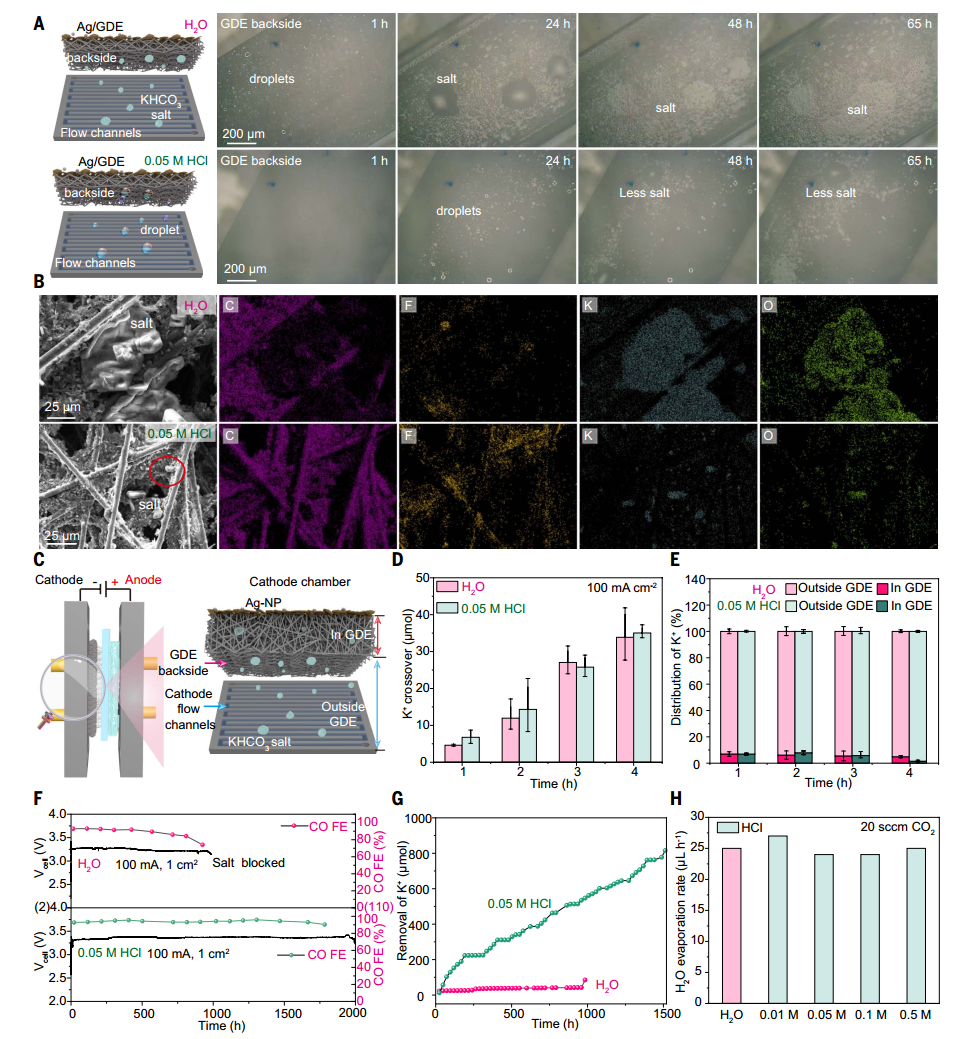
Fig. 2 Mechanism study and salt quantification in CO2RR MEA electrolyzer humidified by H2O and acid
3. Screening the stability of CO2RR for CO2 humidification with various acids
It is found that acid humidification (such as HCl, HCOOH, CH3COOOH) significantly reduces the salt accumulation in CO2RR electrolyzer and prolongs the stability. The experiment shows that when HCl is humidified, the amount of Cl- introduced is related to the concentration, but low concentration (0.05 M) of HCl can effectively remove salt and has no significant effect on the activity and selectivity of CO2RR. Other acids (such as HNO₃ and H₂SO₄) can also reduce salt accumulation, but volatile acids (such as HCOOH and CH₃COOH) perform better, and their stability exceeds 2000 hours. Acid humidification can effectively prevent salt blockage by improving salt solubility and promoting salt discharge. In addition, this method is suitable for a variety of catalysts (such as Ag, Bi₂O₃, ZnO, Cu₂O) and different AEM thicknesses, showing wide applicability and stability, and has no obvious negative impact on catalysts or AEM.
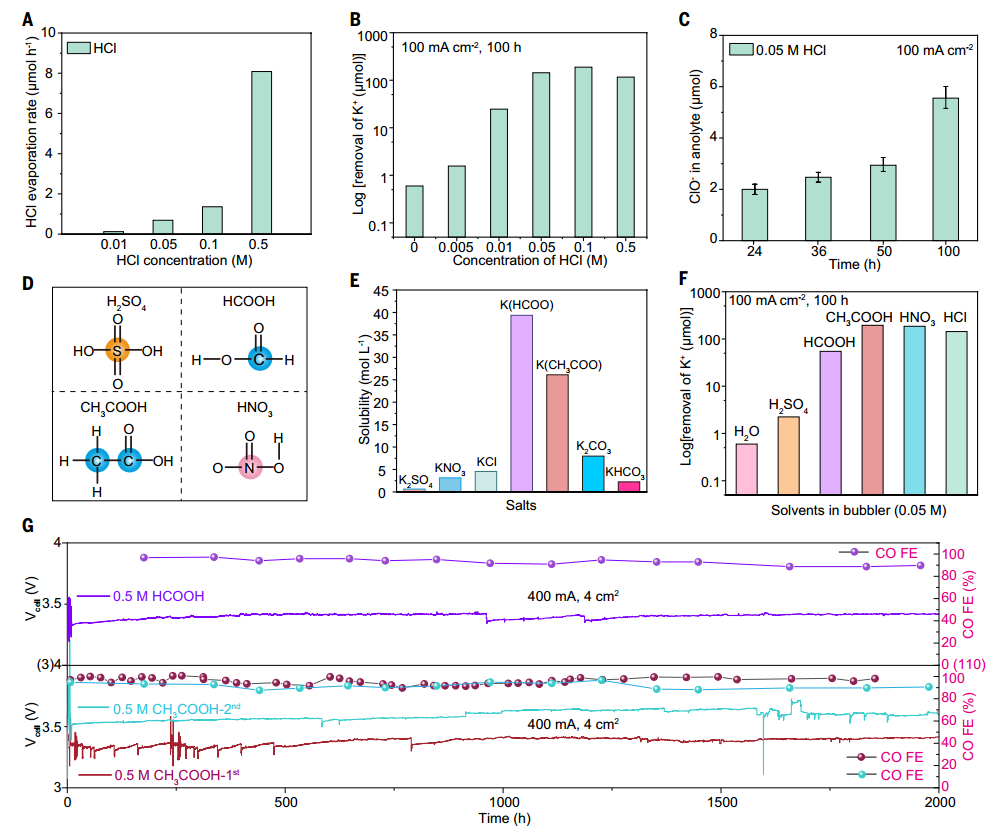
Fig. 3 Effects of different acid concentrations and types on CO2RR electrolysis
4. Stability test of CO2RR to CO in 100 cm2 MEA electrolyzer
The author evaluates the stability of acid humidification method in 100 cm2 CO2RR MEA electrolyzer and finds that it is significantly better than traditional water humidification. In a 100 cm² electrolyzer, the equipment life of water humidification is only about 80 hours, while the CO2 humidified by 0.05 M HCl and 0.05 M HNO₃ has achieved stable operation for more than 2,000 hours and 4,500 hours, respectively. The Faraday efficiency of Co is maintained at 80%-90%, and the battery voltage degradation rate is only about 19 μV h⁻¹. This durability improvement is attributed to the fact that acid humidification effectively prevents the accumulation of KHCO₃ salt in the gas flow channel and the back of GDE. In addition, 0.1 M CH₃COOH humidified co also prolonged the stability to about 1300 hours without salt precipitation. The results also show that the acid humidification method has no significant effect on the morphology of Ag-NP catalyst, and it shows good scalability in large-scale electrolytic cells. Technical and economic analysis shows that the influence of low concentration acid humidification on the overall economic feasibility can be ignored. These results emphasize the effectiveness and economy of acid humidification in large-scale CO2RR electrolyzer, which provides strong support for commercial application.
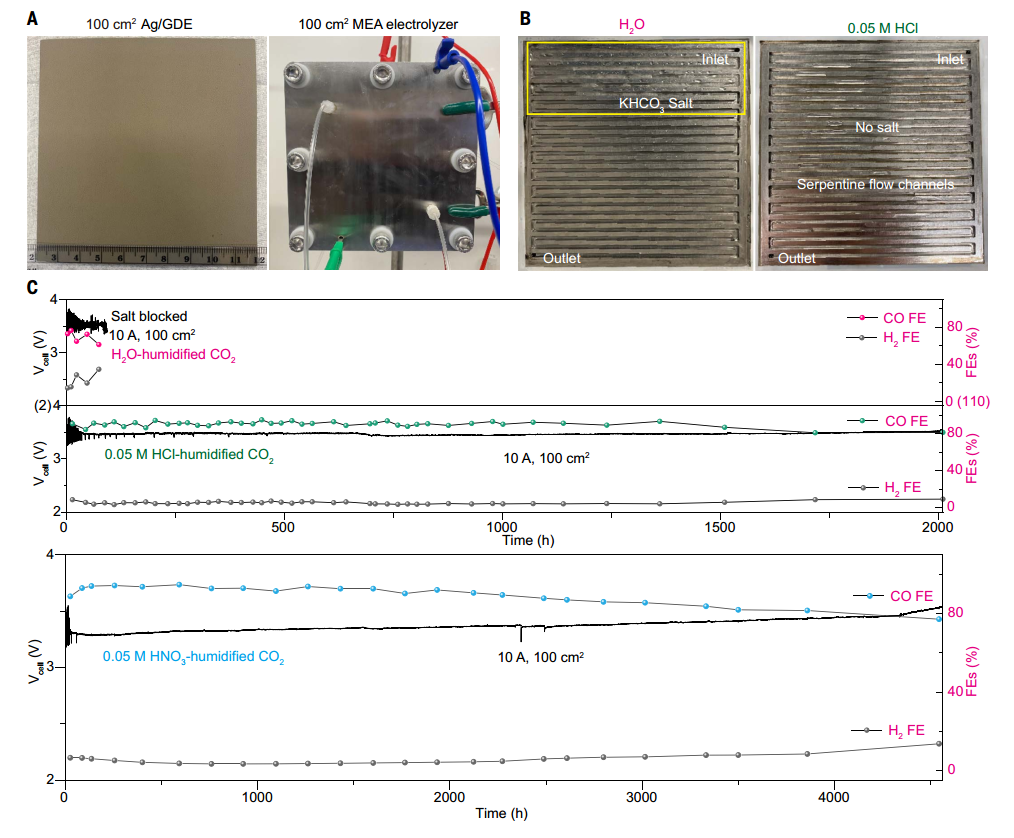
Fig. 4 Stability of CO2RR for humidifying CO2 with H2O and acid in 100cm2 MEA electrolyzer.
Summary and prospect
In a word, the migration of K+ from anolyte to cathode chamber in the process of CO₂RR was deeply discussed in this study, and it was found that the accumulation of bicarbonate formed by it would hinder the mass transfer of CO2 and lead to the failure of electrolytic cell. By humidifying CO2 feed with volatile acid (such as hydrochloric acid), the high solubility of acid and its potassium salt can effectively remove the salt in the cathode chamber and prevent precipitation. This method makes the MEA reactor run stably for more than 4,500 hours, and the CO Faraday efficiency is maintained at 80%-90%, which is obviously superior to the traditional water humidification method, and provides a robust and easy-to-engineer strategy for the commercialization of the CO2RR MEA electrolyzer.
Source:
[1] SHAOYUN HAO, et al. Acid-humidified CO2 gas input for stable electrochemical CO2 reduction reaction. Science, 2025, 388(6752). https://www.science.org/doi/10.1126/science.adr3834
[2] https://mp.weixin.qq.com/s/wIwk4S6WMGL37itzsLyULA
