Recent advances and challenges in electrochemical CO2 reduction to CH4
Background Introduction
Electrochemical reduction (ECR) of CO2 into fuel and basic chemicals by using renewable energy power is an attractive way to reduce CO2 emission and establish carbon neutralization process. Among ECR products, CH4 stands out because of its high calorific value (55.5 MJ kg-1) and clean combustion. However, the slow kinetic process of ECR to produce CH4 involves the 8-proton coupled electron transfer step, which makes the activity and selectivity difficult to control, which is the main challenge for ECR to produce CH4 at present. Therefore, it is urgent to study the systematic reaction mechanism and develop efficient catalysts.
Main innovation points
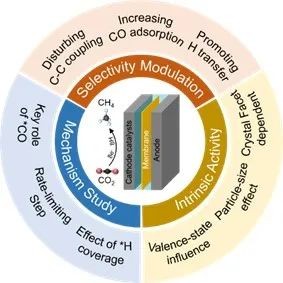
Fig. 1 Schematic diagram of ECR's mechanism research and enhancement strategy for CH4
In this paper, the research progress of CH4 production from ECR in recent years is systematically summarized from the perspective of catalyst design. Firstly, the reaction mechanism of ECR methanation was systematically summarized. Subsequently, the development of ECR catalysts for CH4 production was described in detail, including the influencing factors of intrinsic activity (crystal plane, size and valence state) and the design strategies of high-performance ECR methanation of CH4, such as the development of monoatomic catalysts (SACs), Cu/ oxide interface engineering and Cu surface modification. Finally, this paper puts forward the future challenges and prospects of ECR-catalyzed CH4 production, such as the development of catalysts/devices for industrialization and the development of high-resolution characterization.
Main content
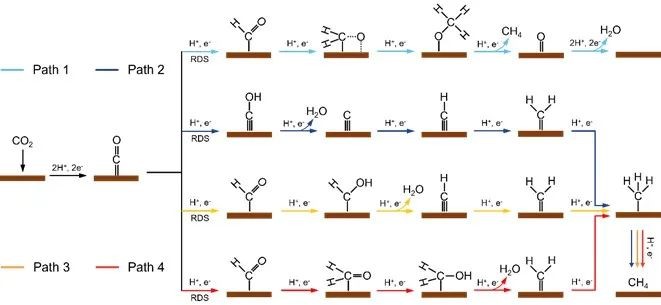
Fig. 2 Reaction mechanism of CH4 production by ECR
Key points:
In this paper, the reaction mechanism of CH4 production from ECR is systematically summarized, which involves eight proton-coupled electron transfer (PCET) processes. CO2 firstly forms an intermediate of *CO through a two-step PCET process, and then further reduces *CO to form CH4. At present, there are three main reaction mechanisms and ways for ECR to produce CH4, and the hydrogenation of *CO to produce hydroxyl formyl (*COH) or formyl (*CHO) has always been considered as one of the most critical steps for ECR to produce CH4.
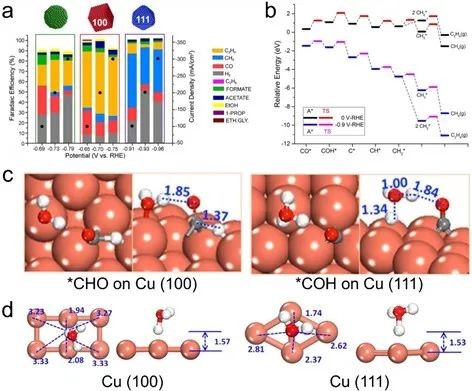
Fig. 3 (a) FE of ECR deep reduction products on (100) and (111) single crystal Cu. (b) At 0 V and -0.9V (vs. RHE), the high potential ECR on the Cu(100) surface produces methane and ethylene through the COH* path. (c) Cu(100) plane water solvation model is used to reduce *CO to *CHO and Cu(111) plane H-shuttle model is used to optimize the transition state of *COH. (d) An example in which *COH on Cu (100) and Cu(111) forms a transition state H3Oσ+
Key points:
Theoretical simulation combined with experimental results show that Cu(111) surface tends to produce *COH by hydrogenation of *CO, and its CH4 selectivity is higher, while Cu(100) is beneficial to the hydrogenation of * CO to produce CHO*, and it is more inclined to produce C2H4 by C-C coupling of two CHO*.
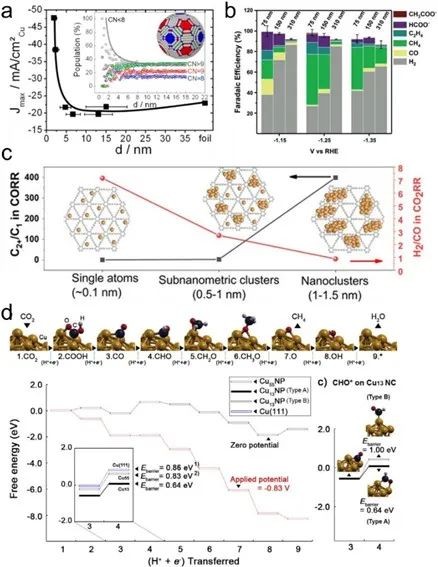
Fig. 4 (a) ECR FE on Cu nanoparticles with particle size ranging from 2 to 15 nm and the function of surface atomic ratio with specific coordination number with particle size. (b) Three sizes of ECR products FE of Cu Oh-NCs. (c) C2+/C1 product ratio of CO reduction on Cu/GDY catalysts with different nano-sizes. (d) The relative free energy diagram of CO2 reduction on Cu55NC and the comparison of potential limiting steps (*CO→*CHO) of Cu13, Cu55 and Cu(111) models
Key points:
The results show that small-sized Cu has better surface flexibility and the center of D-band moves up (closer to Fermi level), which enhances the adsorption and stability of CO2 intermediates including *CHO, reduces the energy barrier of the decisive step of CO2 reduction to CH4, and is more conducive to the selectivity of CH4.
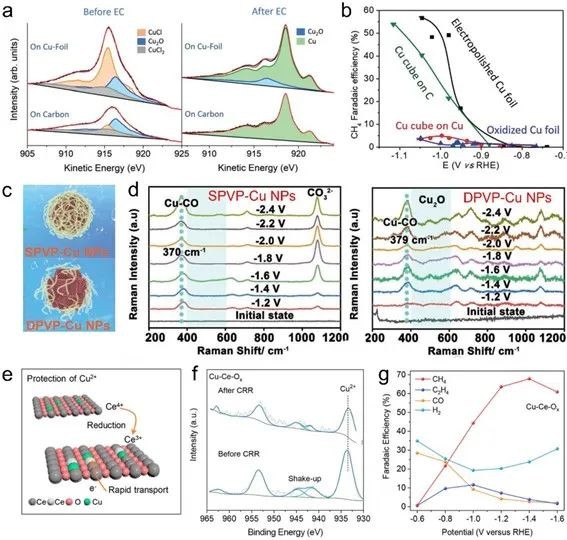
Fig. 5 (a) Quasi-in-situ Cu LMM Auger spectra of Cu nanocubes supported on C paper and copper foil before and after ECR 1h. (b) Schematic diagram of different reminders. (d) In-situ Raman spectra of SPVP-Cu NPs and DPVP-Cu NPs under ECR conditions. (e) Schematic diagram of self-sacrifice mechanism to protect Cu2+. (f) Cu 2p XPS spectrum of Cu-Ce-Ox before and after ECR. (g) Gas product FE of Cu-Ce-Ox at different potentials
Key points:
The valence state of Cu has an opposite conclusion on the selectivity of ECR products, mainly focusing on whether low-priced (such as Cu0) or high-priced (such as Cu2+)Cu has higher selectivity for CH4, so it is necessary to further clarify the influence of valence state effect.
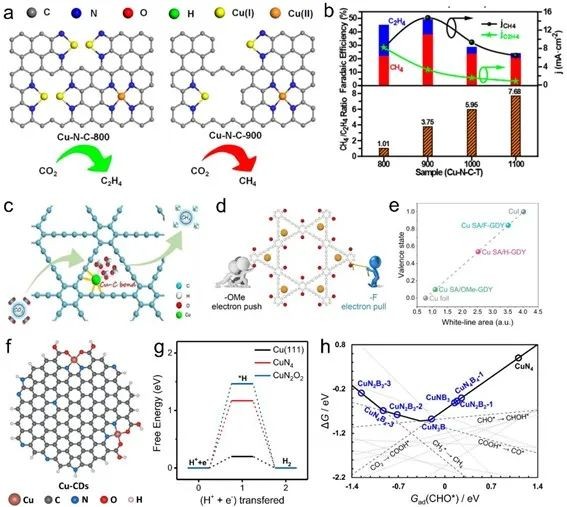
Fig. 6 (a) Schematic diagram of Cu-N-C-T catalysts with different Cu-Cu distances. (b) Faraday efficiency of Cu-N-C-T catalyst, partial current density of CH4 and C2H4 (upper figure, right Y axis) and CH4/C2H4 ratio (lower figure). (c) The diagram of Cu SAs/GDY in which ECR also originated from CH4. (d) electronic push-pull schematic diagram. (e) the average valence state of Cu SA/R-GDY prepared. (f) Cu-N2O2 configuration of Cu-CD catalyst. (g) DFT simulation of hydrogen evolution on CuN2O2 (Cu-CDs), CuN4 and Cu (111). (h) The thermodynamic change of CH4 generated by ECR on Cu-NxBy at -1.2V is related to Gad(CHO*)
Key points:
Cu SACs is a widely reported catalyst for ECR to produce CH4. In order to increase CH4 selectivity, several strategies can be adopted: (1) increasing the distance between adjacent Cu sites (Cu-N4) to prevent C-C coupling; (2) The carrier with stable Cu center (such as graphdiyne, GDY) and even electron-withdrawing group (such as -F) are introduced to improve the valence state of Cu center. (3) Introducing HER nonmetals (such as CuN2O2 and CuB2N2) to improve HER or reduce the reaction barrier of ECR for CH4 production, inhibit her and improve CH4 selectivity.
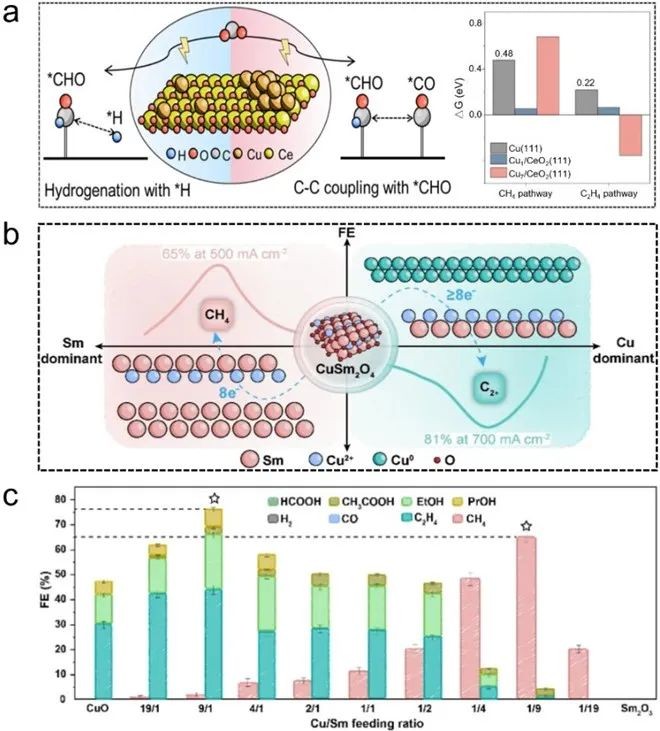
Fig. 7 (a) Schematic diagram of CH4 and C2H4 produced by ECR and free energy of reaction on Cu(111), Cu1/CeO2(111) and Cu7/CeO2(111). (b) Schematic diagram of different proportions of CuSm-Ox regulating ECR to produce C2+ and CH4. (c) Average FE of C2+ and CH4 with different Cu/Sm ratios at 500 mA cm-2
Key points:
Cu/oxide interface can reduce the adsorption energy of CO2 and ECR intermediate (HxCO) and promote the deep reduction of CO2, but different components of Cu/oxide (such as CeO2 and SmO2) have different activities and selectivities for ECR competing products C2+ and CH4. One view is that dispersed Cu species (Cu-Ce-O) in solid solution are active sites for CH4 production, while granular Cu is beneficial for C2+ production. Another viewpoint suggests that the synergism of stable solid solution (such as CuSm2O4) and Cu or oxide phase (Sm2O3) plays a major role in ECR to C2+ and CH4 selectivity, respectively. In a word, higher oxide content will reduce the coordination number of Cu, and use ECR to produce CH4, while higher Cu content has high coordination Cu, which is beneficial to C-C coupling to produce C2+.
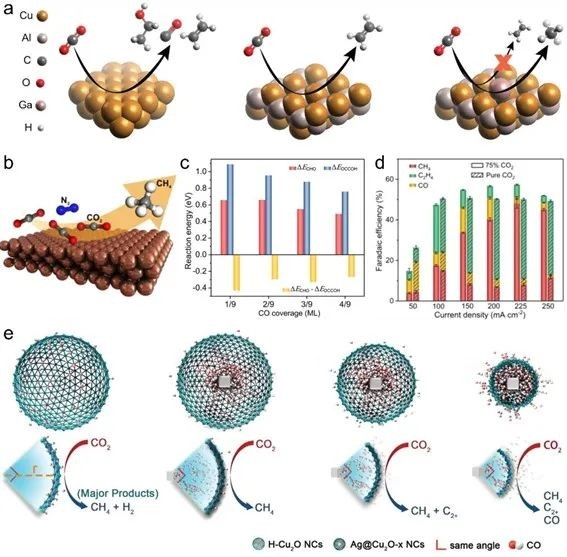
Fig. 8 (a) Doped metal is introduced to inhibit C-C coupling and promote ECR methanation. (b) Schematic diagram of CO2 dilution strategy for adjusting ECR selectivity to CH4. (c) Under different coverage of *CO, *CO protonated into *CHO and C-C coupling reaction barriers. (d) Distribution of ECR gas products at different current densities when pure CO2 and CO2 concentration are 75%. (e) The flux of *C in Ag@Cu2O-xNC with different OD-Cu coating sizes, and the regulation of divergent *CO molecules generated from Ag core on the reduction products
Key points:
There is a competitive relationship between C2+ products produced by coupling intermediates (such as *CO) and CH4 produced by hydrogenation, which will greatly reduce the selectivity of CH4 produced by ECR. By introducing elements with low *CO adsorption energy (such as Ga) and reducing *CO coverage (such as diluting CO2 concentration and increasing the reaction space of Cu site), C-C coupling can be inhibited, and the hydrogenation of *CO to CH4 can be promoted.
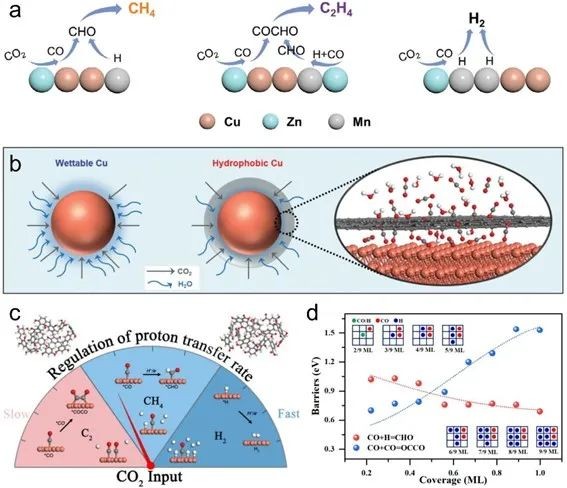
Fig. 9. (a) Schematic diagram of CH4, C2H4 and H2 product distribution with different *CO and *H coverage on CuxZnyMnz catalyst. (b) Schematic diagram of hydrophobic core-shell structure, which can limit the availability of water through hydrophobic carbon coating. (c) Schematic diagram of adjusting proton transfer kinetics on C2+ or CH4 to control ECR products. (d) Energy barrier of hydrogenation and coupling of *CO under different hydrogen coverage
Key points:
ECR to CH4 involves 8 proton transfer, and its decisive step involves PCET process, while C2+ path decisive step is not PCET. With the increase of proton activation and transfer rate, proton-coupled electron transfer is dominant, which can promote the hydrogenation of *CO to CH4. If the proton activation and transfer rate is further increased, HER reaction will be dominant, but Faraday efficiency of CH4 will be inhibited.
Summary
This paper systematically summarizes and discusses the current research progress and future application strategy of CH4 production by ECR.
1. The reaction mechanism of CH4 production from ECR is deeply discussed, including three recognized reaction pathways, involving three key intermediates, namely *CO, *CHO (or *COH) and * H. The decisive step of the reaction is the first hydrogenation of *CO, namely * CO+H+E-→ * CHO (or *COH).
2. According to the key copper-based catalyst for CH4 production from ECR, the key factors affecting its intrinsic activity were summarized from the aspects of crystal plane, size and valence state. Among them, (111) crystal plane and small size are favorable for CH4 production, but the effect of valence states of Cu (Cu0 and Cux+) on ECR selectivity is still controversial.
3. The recent design strategies of ECR methanation catalyst are summarized, including sub-nano catalyst development, Cu/ oxide interface engineering and Cu surface modification, which are mainly based on three principles: (1) regulating the adsorption strength of key intermediate (*CO), such as electrolyte ion influence, valence state regulation and interface engineering; (2) inhibiting C-C coupling, such as reducing the number of active sites and the coverage of intermediate (*CO); (3) moderate *H species activation and transfer.
4. Based on the high-resolution in-situ characterization method, efficient reactor design and Qualcomm screening method, the future development of efficient conversion of ECR to CH4 is prospected.
Source:
[1] Lei Xiong, Xianbiao Fu*,Chin. J. Catal., 2025, 73, 39-61.
[2] https://mp.weixin.qq.com/s/BfxIJyjhwZD5oyXh5uryiw
